The Top 21 Supplements For Superhuman Energy
The Big Key To REAL Energy Enhancement Isn’t Sugar and Stimulants – It’s Optimizing Your Mitochondria!
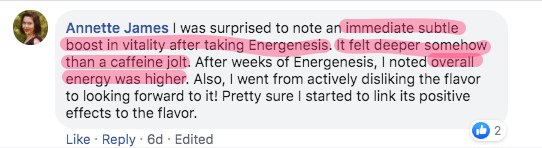
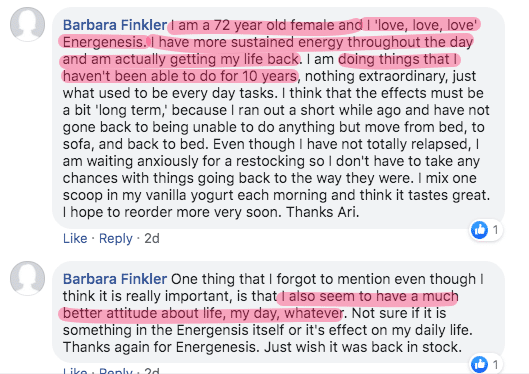
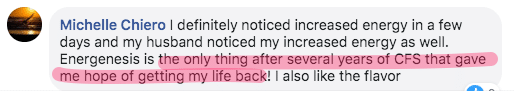
What people say about Energenesis





Worldwide Shipping (FREE Shipping within the U.S.)*
* For international orders: All shipments are shipped Duties, Customs, & Taxes unpaid. Buyer is responsible for delivery fees.
What's In Energenesis
Energenesis is loaded with 23 of the highest mitochondria-building ingredients in existence.
Energenesis is like a 5-star MEAL for your Mitochondria (LOADED with rare nutrients, energy boosters and powerful antioxidants, because your body needs a variety – each one works differently!) AND… we have traveled the globe to provide the highest ingredients, with unparalleled potency, purity, biological availability, and effectiveness…
Mitochondrial Membrane Support Complex
NTFactor® Phospholipid Complex
NTFactor® is a digestion-resistant phospholipid complex that supplies the materials necessary to repair and regenerate damaged cell membranes directly to mitochondria, a treatment called lipid replacement therapy [1].
By supplying the bioactive phospholipids that mitochondria require to naturally replace damaged membrane components, NTFactor® is able to regenerate dysfunctional mitochondria, increase energy production, and enhance the energy levels of those using it.
Numerous studies have shown that NTFactor® reduces fatigue by 24–43 percent among those with chronic fatigue syndrome or conditions associated with fatigue, like general aging, obesity, lyme disease, and Gulf War Illness [2]. In older adults, NTFactor® restored mitochondrial function to a level matching healthy 29 year-olds [3].
Astaxanthin
Astaxanthin is one of the most powerful membrane-specific antioxidants in existence. Not only is it 30–85 percent more potent than vitamin E and carotenoids like β-carotene, lycopene, and lutein [4], but it has a unique polar structure that allows it to cross through, protect, and stabilize the entire mitochondrial membrane [5].
Due to astaxanthin’s consistent and powerful ability to prevent oxidative damage and dysfunction to mitochondria [6–8], some researchers have even determined it to be “mitochondria-targeted antioxidant” [9].
Interventions have shown that astaxanthin supplementation increases muscular strength and power output in fatigued adults [10], enhances muscular endurance and recovery in recreational athletes [11,12], and reduces muscle damage and oxidative stress in elite soccer players [13].
Mitochondrial Energy Production Cofactor Complex
Acetyl-L-Carnitine
Acetyl-L-Carnitine (ALCAR) is a special form of carnitine that achieves two goals: (1) it supplies the carnitine your mitochondria need to produce energy, and (2) it provides an acetyl moiety that your mitochondria use to remain youthful and healthy.
The carnitine shuttle system is essential for bringing fatty acids into mitochondria to be used for energy production, and carnitine deficits (even mild ones) have been documented in those with chronic fatigue [14]. Moreover, over 20 percent of mitochondrial proteins rely on acetyl moieties to properly function, including those involved in antioxidant defenses and energy production [15,16].
For these reasons, some researchers have proposed that ALCAR should be considered a “mitochondrial rejuvenator” [17]. In chronically fatigued older adults, ALCAR supplementation led to profound benefits to their wellbeing — a 24 percent increase in physical function and close to a whopping 50 percent reduction in mental fatigue, physical fatigue, and overall fatigue severity [18].
D-Ribose
D-ribose is a naturally occurring sugar molecule that assists in the production of cellular energy by virtue of being a necessary component of ATP, DNA, and RNA [19]. In particular, it is the product of a rate-limiting step in ATP production within mitochondria and supplementation directly bypasses this bottleneck to increase energy production [20].
Studies show that D-ribose can help boost energy and physical function in situations where energy levels are reduced, such as people who have suffered from heart disease or stroke [21–23], or people engaging in regular intense exercise [24,25].
In adults with chronic fatigue, supplementing D-ribose led to 45 percent greater energy levels, 25 percent better sleep quality, 16% more mental clarity, 14 percent less pain, and 30% greater overall wellbeing [26]. Another study reported similar findings, with all benefits disappearing within a week of stopping supplementation [27].
Creatine
Creatine is a molecule necessary for rapid and explosive energy production when energy needs cannot be immediately met by our mitochondria. Supplementation works by increasing the amount of available creatine within our muscles.
This not only increases muscular performance, but also helps stimulate mitochondrial energy production when faced with signals to do so (like being physically active) [28], enhance mitochondrial biogenesis and integrity [29], and protect mitochondria from oxidative damage [30,31].
Accordingly, creatine is one of the most extensively researched supplements for improving exercise performance, muscle strength, and power output in people of all ages [32–36].
Magnesium
Magnesium is required for over 300 enzymes to function properly — muscle contractions, blood vessel regulation, insulin signaling, the synthesis of DNA and proteins, and nerve transmission all require magnesium [37].
One of the enzymes that relies on magnesium is ATP synthase, which is responsible for generating cellular energy (ATP) within mitochondria. Without magnesium, your mitochondria simply can’t function efficiently [38,39].
Energenesis provides two forms of magnesium, citrate and malate, which are both cofactors for mitochondrial energy production themselves.
Coenzyme Q10
Coenzyme Q10 (CoQ10) is an essential component of mitochondrial energy production, serving as both an antioxidant and an energy-transferring molecule. As such, deficits in CoQ10 will not only lead to a cessation of energy production, but also an increase in oxidative damage.
Individuals with chronic fatigue regularly show deficiencies in CoQ10 concentrations throughout the body [40], as do those with conditions in which fatigue is a common symptom, like fibromyalgia [41–43], those who have survived heart attacks or heart failure [44,45], and multiple sclerosis [46,47].
Supplementation with CoQ10 improves fatigue, autonomic nervous system activity (the part of the nervous system involved in rest and recovery), and biochemical parameters of mitochondrial energy production in those with chronic fatigue [48–51], and reduces general fatigue and oxidative stress in healthy adults [52,53].
Mitochondrial Biogenesis and Defense Complex
Green Tea Catechins
Green tea (Camellia Sinensis) catechins are four phytochemical molecules, the most potent one being epigallocatechin-3-gallate (EGCG). It has been implicated in benefiting almost every organ system in the body in doses you can obtain easily from simply drinking green tea [54–56].
EGCG is neuroprotective [57,58], cardioprotective [59,60], anti-obesity [61–63], anti-carcinogenic [64,65], and anti-diabetic [66], all due primarily to its ability to stimulate mitochondrial biogenesis, enhance energy production, and protect mitochondria from oxidative stress [67,68].
Supplementation of EGCG plus resveratrol (another ingredient of Energenesis) has been shown to significantly increase mitochondrial function and the use of fatty acids for energy production [69], and several meta-analyses have concluded that EGCG reduces body weight and body fat [70–73], particularly abdominal fat [74].
Pomegranate
Pomegranate is a rich source of ellagitannins, potent antioxidant molecules that can be further metabolized into other antioxidant derivatives like ellagic acid and urolithins [75,76]. These substances have been heavily investigated for their mitochondrial and cardiovascular benefits.
Pomegranate ellagitannins and their derivatives have been shown to enhance mitochondrial function [77] and increase rates of mitophagy (mitochondria + autophagy) [78,79], which is a quality control pathway that preserves mitochondrial health by targeting damaged mitochondria for autophagic degradation, making anything that facilitates mitophagy absolutely vital for optimal health and disease prevention.
In recreational endurance athletes, supplementing with pomegranate extract for just two weeks increased the total time the athletes could cycle before complete exhaustion by 14 percent and increased the amount of time they could rely on their mitochondria to supply most of their energy by 10 percent [80].
Moreover, pomegranate supplementation has been shown to reduce blood lipid oxidation and the accumulation of plaque in arteries [81], particularly in people who have higher levels of oxidative stress [82,83].
Panax Ginseng
Panax ginseng has been used medicinally for thousands of years in China, Korea, and Japan to alleviate physical and mental fatigue. While there are several types of ginseng on the market, panax ginseng is considered the “true” ginseng.
At a fundamental level, panax ginseng works to protect mitochondria from oxidative damage and improve energy production under conditions of oxidative stress [84,85]. A systematic review and meta-analysis of five studies in chronic fatigue patients found significant benefits of ginseng supplementation for reducing fatigue severity [86], with reductions of 20 percent after one month not being uncommon [87,88].
Ashwagandha
Ashwagandha (Withania somnifera) is a nightshade revered in Ayurvedic medicine for its physical- and mental-enhancing effects [89]. Today, it’s considered an adaptogen for similar reasons, able to increase a person’s resilience to stress and help reduce anxiety [90,91].
These effects are largely due to its constituent withanolide structures, which have several important neuroprotective effects within the brain, such as scavenging free radicals, reducing neuroinflammation, and promoting neurotransmitter signaling [92]. They also improve mitochondrial function and energy production [93].
In Energenesis, we use the most potent form of ashwagandha, a 35% withanolide concentrate called the Shoden extract. Just 240 mg of Shoden ashwagandha reduces stress and anxiety by 30% in mildly stressed adults [94], while a mere 60 mg can boost DHEA-S and testosterone by 15–18% in middle-aged men
In another large randomized placebo-controlled trial of 150 men and women, supplementing 120 mg has been documented to improve physical and psychological quality of life by 8% and enhance sleep quality by 40% compared to placebo, including dramatically improving the time it took to fall asleep, total sleep time, and nightly awakenings (Deshpande et al. 2020).
Other studies have shown ashwagandha supplementation to reduce perceived stress, anxiety, and depression in mildly stressed healthy adults [95,96], adults battling chronic mental stress [97], and adults with an anxiety disorder [98].
Alpha-Lipoic Acid
Alpha-lipoic acid (ALA) is a mitochondrial molecule involved in energy metabolism and the antioxidant system. It is not only essential for mitochondria to create cellular energy, but also serves to function as an antioxidant, replenish other antioxidants, and stimulate the production of antioxidant enzymes like glutathione [99].
Accordingly, ALA has been heavily investigated as a mitochondrial rejunivator, able to help reverse age-related declines in mitochondrial energy production [100], particularly within the brain [101,102].
By improving mitochondrial function and protecting against oxidative stress, ALA has been shown to benefit metabolic health [103,104], as well as facilitate weight loss among those who are overweight [105–107].
ALA exists as either an S or R isomer, with unspecified ALA being a 50-50 ‘racemic’ solution of both. Most studies use the racemic solution, but only R-ALA has biological activity in the body and is more bioavailable [108], which is why we use it in Energenesis.
Pyrroloquinoline Quinone
Pyrroloquinoline Quinone (PQQ) is a potent stimulator of pathways involved in mitochondrial biogenesis and antioxidant defenses [109,110]. In particular, it stimulates pathways shared by exercise training and is believed to potentiate and enhance activity-induced benefits on mitochondria [111].
Supplementation has been shown to reduce fatigue and increase vigor by 20 percent in adults complaining of poor sleep and energy levels, as well as improve mood, sleep quality, and overall quality of life [112]. In another study of healthy adults, PQQ reduced inflammation and improved markers of mitochondrial respiration [113].
NAD+ Regeneration Complex
Taurine
Taurine is an omnipresent amino acid within the body, essential for the development and function of our cardiovascular, muscular, nervous, and ocular systems [114]. It’s also essential for mitochondrial energy production and protein synthesis [115,116]
The highest amounts of taurine are in tissues with huge energy requirements and a lot of mitochondria, such as the retina, nerves, kidney, heart, and skeletal muscle [117]. If mitochondria don’t have enough taurine, energy production decreases and oxidative stress increases [118].
Insufficient taurine has been implicated in numerous chronic disease states [119,120], and several clinical trials have shown that supplementing with taurine reduces oxidative stress and inflammation in those with metabolic dysfunction [121–123].
Additionally, a meta-analysis reported that taurine supplementation improved endurance exercise performance, particularly power output and the amount of time that people could run before exhaustion [124].
Citrus Bioflavonoids
Citrus bioflavonoids are a group of molecules, particularly hesperidin, naringenin, and rutin, which have potent antioxidant and anti-inflammatory effects within the body [125]. They also increase mitochondrial energy production, biogenesis, and mitophagy [126–129].
Supplementation has been shown to increase sprint performance, power output, and exercise economy (requiring less energy to generate a given power output) in athletes [130,131].
Resveratrol
Resveratrol is a phytochemical found primarily in grape skins and wine. It works primarily through hormesis [132]; it stresses our mitochondria and stimulates adaptations that ultimately make them bigger and stronger [133].
Supplementation has been shown to activate several mitochondrial regulators, including AMPK, SIRT1, and PGC-1α, while also increasing the ability of mitochondria to oxidize fat as an energy source [134,135]. Resveratrol also increases mitochondrial adaptations to exercise more than exercise alone would [136,137].
Quercetin
Quercetin is a potent antioxidant and anti-inflammatory molecule that affects an array of mitochondrial processes, including mitochondrial biogenesis, mitochondrial energy production, and the protection of mitochondria from oxidative stress [138,139].
Several meta-analyses of clinical trials have reported that quercetin is able to improve endurance exercise performance and maximal oxygen consumption [140,141], and, in those with metabolic dysfunction, reduce markers of inflammation [142,143], improve blood lipids [144–146], and lower blood pressure [147,148].
Importantly, these all use regular unenhanced quercetin. Yet, a quercetin phytosome complex has vastly superior bioavailability, leading to quercetin levels 20-fold greater in the blood following supplementation [149], which is why we use this form in Energenesis.
N-Acetyl-Cysteine
N-Acetyl-Cysteine (NAC) is a molecule our body naturally creates from the amino acid cysteine as a precursor to one of our body’s master antioxidants, glutathione, which is not only a potent antioxidant, but a molecule required to recycle other antioxidants, detoxify and excrete toxins, and maintain mitochondrial function [150].
Supplemental NAC is an effective way at increasing glutathione concentrations in our body [151,152], and a meta-analysis of 28 clinical trials found that NAC supplementation significantly reduced biomarkers of oxidative stress and inflammation [153].
Through helping to sustain optimal glutathione concentrations, NAC has been demonstrated to reduce mitochondrial oxidative damage and preserve cellular life in the face of genetic mitochondrial mutations that cause dysfunction or toxic conditions that directly damage mitochondria [154–157].
Niacinamide
Niacin is an essential component of energy-carrying molecules within the mitochondria, all of which are derivatives of nicotinamide adenine dinucleotide (NAD+). NAD+ is important for energy production, mitochondrial function, and cellular repair processes.
The primary route of NAD+ synthesis is through something called the salvage pathway, in which niacinamide serves as the starting molecule. We can also make NAD+ from nicotinic acid using an alternative pathway, which is why we’ve included both forms in Energenesis to ensure that NAD+ generation is not being impeded by a simple vitamin insufficiency.
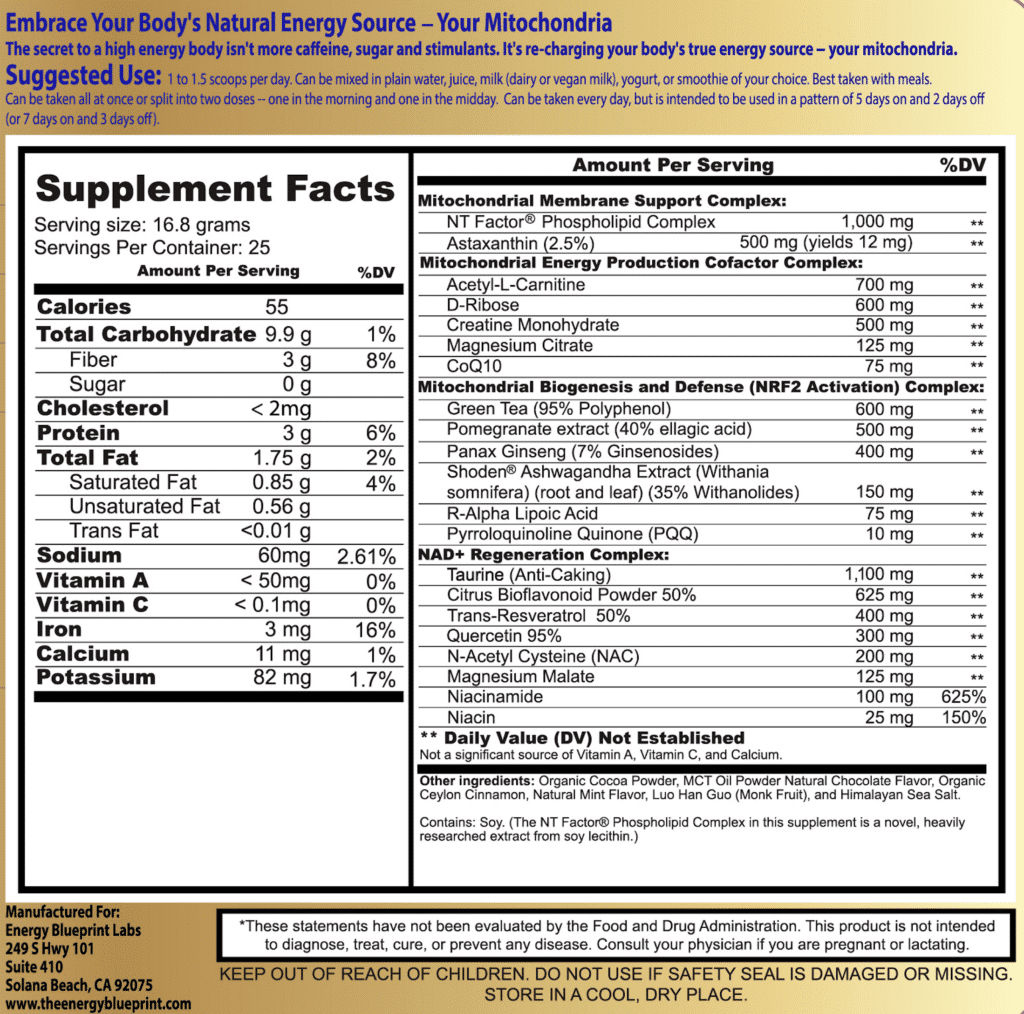
If you want to see a breakdown of the exact ingredients and amounts, you can look in the right column of the image below…
What people say about Energenesis





Your cells need a powerful recipe for energy, and Energenesis is it.
Worldwide Shipping (FREE Shipping within the U.S.)*
* For international orders: All shipments are shipped Duties, Customs, & Taxes unpaid. Buyer is responsible for delivery fees.
References
- Nicolson GL, Ash ME. Lipid Replacement Therapy: a natural medicine approach to replacing damaged lipids in cellular membranes and organelles and restoring function. Biochim Biophys Acta. 2014;1838:1657–79.
- Nicolson GL, Rosenblatt S, de Mattos GF, Settineri R, Breeding PC, Ellithorpe RR, et al. Clinical Uses of Membrane Lipid Replacement Supplements in Restoring Membrane Function and Reducing Fatigue in Chronic Diseases and Cancer. Discoveries (Craiova). 2016;4:e54.
- Agadjanyan M, Vasilevko V, Ghochikyan A, Berns P, Kesslak P, Settineri RA, et al. Nutritional Supplement (NT FactorTM) Restores Mitochondrial Function and Reduces Moderately Severe Fatigue in Aged Subjects. J Chronic Fatigue Syndr. Taylor & Francis; 2003;11:23–36.
- Naguib YM. Antioxidant activities of astaxanthin and related carotenoids. J Agric Food Chem. 2000;48:1150–4.
- Kidd P. Astaxanthin, cell membrane nutrient with diverse clinical benefits and anti-aging potential. Altern Med Rev. 2011;16:355–64.
- Kim SH, Kim H. Inhibitory Effect of Astaxanthin on Oxidative Stress-Induced Mitochondrial Dysfunction-A Mini-Review. Nutrients [Internet]. 2018;10. Available from: http://dx.doi.org/10.3390/nu10091137
- Yu T, Dohl J, Chen Y, Gasier HG, Deuster PA. Astaxanthin but not quercetin preserves mitochondrial integrity and function, ameliorates oxidative stress, and reduces heat-induced skeletal muscle injury. J Cell Physiol. 2019;234:13292–302.
- Krestinina O, Baburina Y, Krestinin R, Odinokova I, Fadeeva I, Sotnikova L. Astaxanthin Prevents Mitochondrial Impairment Induced by Isoproterenol in Isolated Rat Heart Mitochondria. Antioxidants (Basel) [Internet]. 2020;9. Available from: http://dx.doi.org/10.3390/antiox9030262
- Sztretye M, Dienes B, Gönczi M, Czirják T, Csernoch L, Dux L, et al. Astaxanthin: A Potential Mitochondrial-Targeted Antioxidant Treatment in Diseases and with Aging. Oxid Med Cell Longev. 2019;2019:3849692.
- Liu SZ, Ali AS, Campbell MD, Kilroy K, Shankland EG, Roshanravan B, et al. Building strength, endurance, and mobility using an astaxanthin formulation with functional training in elderly. J Cachexia Sarcopenia Muscle. 2018;9:826–33.
- Malmsten CL, Lignell A. Dietary Supplementation with Astaxanthin-Rich Algal Meal Improves Strength Endurance–A Double Blind Placebo Controlled Study on Male Students–. Carotenoid Sci. 2008;13:20–2.
- Fleischmann C, Horowitz M, Yanovich R, Raz H, Heled Y. Asthaxanthin Improves Aerobic Exercise Recovery Without Affecting Heat Tolerance in Humans. Front Sports Act Living. 2019;1:17.
- Djordjevic B, Baralic I, Kotur-Stevuljevic J, Stefanovic A, Ivanisevic J, Radivojevic N, et al. Effect of astaxanthin supplementation on muscle damage and oxidative stress markers in elite young soccer players. J Sports Med Phys Fitness. 2012;52:382–92.
- Filler K, Lyon D, Bennett J, McCain N, Elswick R, Lukkahatai N, et al. Association of Mitochondrial Dysfunction and Fatigue: A Review of the Literature. BBA Clin. 2014;1:12–23.
- Kim SC, Sprung R, Chen Y, Xu Y, Ball H, Pei J, et al. Substrate and functional diversity of lysine acetylation revealed by a proteomics survey. Mol Cell. 2006;23:607–18.
- Kerner J, Yohannes E, Lee K, Virmani A, Koverech A, Cavazza C, et al. Acetyl-L-carnitine increases mitochondrial protein acetylation in the aged rat heart. Mech Ageing Dev. 2015;145:39–50.
- Rosca MG, Lemieux H, Hoppel CL. Mitochondria in the elderly: Is acetylcarnitine a rejuvenator? Adv Drug Deliv Rev. 2009;61:1332–42.
- Malaguarnera M, Gargante MP, Cristaldi E, Colonna V, Messano M, Koverech A, et al. Acetyl L-carnitine (ALC) treatment in elderly patients with fatigue. Arch Gerontol Geriatr. 2008;46:181–90.
- Pauly DF, Pepine CJ. D-Ribose as a supplement for cardiac energy metabolism. J Cardiovasc Pharmacol Ther. 2000;5:249–58.
- Mahoney DE, Hiebert JB, Thimmesch A, Pierce JT, Vacek JL, Clancy RL, et al. Understanding D-Ribose and Mitochondrial Function. Adv Biosci Clin Med. 2018;6:1–5.
- Omran H, Illien S, MacCarter D, St Cyr J, Lüderitz B. D-Ribose improves diastolic function and quality of life in congestive heart failure patients: a prospective feasibility study. Eur J Heart Fail. 2003;5:615–9.
- MacCarter D, Vijay N, Washam M, Shecterle L, Sierminski H, St Cyr JA. D-ribose aids advanced ischemic heart failure patients. Int J Cardiol. 2009;137:79–80.
- Pliml W, von Arnim T, Stäblein A, Hofmann H, Zimmer HG, Erdmann E. Effects of ribose on exercise-induced ischaemia in stable coronary artery disease. Lancet. 1992;340:507–10.
- Hellsten Y, Skadhauge L, Bangsbo J. Effect of ribose supplementation on resynthesis of adenine nucleotides after intense intermittent training in humans. Am J Physiol Regul Integr Comp Physiol. 2004;286:R182–8.
- Seifert JG, Brumet A, St Cyr JA. The influence of D-ribose ingestion and fitness level on performance and recovery. J Int Soc Sports Nutr. 2017;14:47.
- Teitelbaum JE, Johnson C, St Cyr J. The use of D-ribose in chronic fatigue syndrome and fibromyalgia: a pilot study. J Altern Complement Med. 2006;12:857–62.
- Gebhart B, Jorgenson JA. Benefit of ribose in a patient with fibromyalgia. Pharmacotherapy. 2004;24:1646–8.
- Walsh B, Tonkonogi M, Söderlund K, Hultman E, Saks V, Sahlin K. The role of phosphorylcreatine and creatine in the regulation of mitochondrial respiration in human skeletal muscle. J Physiol. 2001;537:971–8.
- Barbieri E, Guescini M, Calcabrini C, Vallorani L, Diaz AR, Fimognari C, et al. Creatine Prevents the Structural and Functional Damage to Mitochondria in Myogenic, Oxidatively Stressed C2C12 Cells and Restores Their Differentiation Capacity. Oxid Med Cell Longev. 2016;2016:5152029.
- Sestili P, Barbieri E, Martinelli C, Battistelli M, Guescini M, Vallorani L, et al. Creatine supplementation prevents the inhibition of myogenic differentiation in oxidatively injured C2C12 murine myoblasts. Mol Nutr Food Res. 2009;53:1187–204.
- Sestili P, Barbieri E, Stocchi V. Effects of Creatine in Skeletal Muscle Cells and in Myoblasts Differentiating Under Normal or Oxidatively Stressing Conditions. Mini Rev Med Chem. 2016;16:4–11.
- Dempsey RL, Mazzone MF, Meurer LN. Does oral creatine supplementation improve strength? A meta-analysis. J Fam Pract. 2002;51:945–51.
- Branch JD. Effect of creatine supplementation on body composition and performance: a meta-analysis. Int J Sport Nutr Exerc Metab. 2003;13:198–226.
- Lanhers C, Pereira B, Naughton G, Trousselard M, Lesage F-X, Dutheil F. Creatine Supplementation and Lower Limb Strength Performance: A Systematic Review and Meta-Analyses. Sports Med. 2015;45:1285–94.
- Lanhers C, Pereira B, Naughton G, Trousselard M, Lesage F-X, Dutheil F. Creatine Supplementation and Upper Limb Strength Performance: A Systematic Review and Meta-Analysis. Sports Med. 2017;47:163–73.
- Chilibeck PD, Kaviani M, Candow DG, Zello GA. Effect of creatine supplementation during resistance training on lean tissue mass and muscular strength in older adults: a meta-analysis. Open Access J Sports Med. 2017;8:213–26.
- Swaminathan R. Magnesium metabolism and its disorders. Clin Biochem Rev. 2003;24:47–66.
- Igamberdiev AU, Kleczkowski LA. Optimization of ATP synthase function in mitochondria and chloroplasts via the adenylate kinase equilibrium. Front Plant Sci. 2015;6:10.
- Pilchova I, Klacanova K, Tatarkova Z, Kaplan P, Racay P. The Involvement of Mg2+ in Regulation of Cellular and Mitochondrial Functions. Oxid Med Cell Longev. 2017;2017:6797460.
- Filler K, Lyon D, Bennett J, McCain N, Elswick R, Lukkahatai N, et al. Association of Mitochondrial Dysfunction and Fatigue: A Review of the Literature. BBA Clin. 2014;1:12–23.
- Cordero MD, Moreno-Fernández AM, deMiguel M, Bonal P, Campa F, Jiménez-Jiménez LM, et al. Coenzyme Q10 distribution in blood is altered in patients with fibromyalgia. Clin Biochem. 2009;42:732–5.
- Di Pierro F, Rossi A, Consensi A, Giacomelli C, Bazzichi L. Role for a water-soluble form of CoQ10 in female subjects affected by fibromyalgia. A preliminary study. Clin Exp Rheumatol. 2017;35 Suppl 105:20–7.
- Cordero MD, Alcocer-Gómez E, de Miguel M, Culic O, Carrión AM, Alvarez-Suarez JM, et al. Can coenzyme q10 improve clinical and molecular parameters in fibromyalgia? Antioxid Redox Signal. 2013;19:1356–61.
- Jafari M, Mousavi SM, Asgharzadeh A, Yazdani N. Coenzyme Q10 in the treatment of heart failure: A systematic review of systematic reviews. Indian Heart J. 2018;70 Suppl 1:S111–7.
- DiNicolantonio JJ, Bhutani J, McCarty MF, O’Keefe JH. Coenzyme Q10 for the treatment of heart failure: a review of the literature. Open Heart. 2015;2:e000326.
- Sanoobar M, Dehghan P, Khalili M, Azimi A, Seifar F. Coenzyme Q10 as a treatment for fatigue and depression in multiple sclerosis patients: A double blind randomized clinical trial. Nutr Neurosci. 2016;19:138–43.
- Sanoobar M, Eghtesadi S, Azimi A, Khalili M, Khodadadi B, Jazayeri S, et al. Coenzyme Q10 supplementation ameliorates inflammatory markers in patients with multiple sclerosis: a double blind, placebo, controlled randomized clinical trial. Nutr Neurosci. 2015;18:169–76.
- Castro-Marrero J, Cordero MD, Segundo MJ, Sáez-Francàs N, Calvo N, Román-Malo L, et al. Does oral coenzyme Q10 plus NADH supplementation improve fatigue and biochemical parameters in chronic fatigue syndrome? Antioxid Redox Signal. 2015;22:679–85.
- Fukuda S, Nojima J, Kajimoto O, Yamaguti K, Nakatomi Y, Kuratsune H, et al. Ubiquinol-10 supplementation improves autonomic nervous function and cognitive function in chronic fatigue syndrome. Biofactors. 2016;42:431–40.
- Mizuno K, Tanaka M, Nozaki S, Mizuma H, Ataka S, Tahara T, et al. Antifatigue effects of coenzyme Q10 during physical fatigue. Nutrition. 2008;24:293–9.
- Castro-Marrero J, Sáez-Francàs N, Segundo MJ, Calvo N, Faro M, Aliste L, et al. Effect of coenzyme Q10 plus nicotinamide adenine dinucleotide supplementation on maximum heart rate after exercise testing in chronic fatigue syndrome – A randomized, controlled, double-blind trial. Clin Nutr. 2016;35:826–34.
- Mizuno K, Sasaki AT, Watanabe K, Watanabe Y. Ubiquinol-10 Intake Is Effective in Relieving Mild Fatigue in Healthy Individuals. Nutrients [Internet]. 2020;12. Available from: http://dx.doi.org/10.3390/nu12061640
- Sarmiento A, Diaz-Castro J, Pulido-Moran M, Moreno-Fernandez J, Kajarabille N, Chirosa I, et al. Short-term ubiquinol supplementation reduces oxidative stress associated with strenuous exercise in healthy adults: A randomized trial. Biofactors. 2016;42:612–22.
- Singhal K, Raj N, Gupta K, Singh S. Probable benefits of green tea with genetic implications. J Oral Maxillofac Pathol. 2017;21:107–14.
- Suzuki Y, Miyoshi N, Isemura M. Health-promoting effects of green tea. Proc Jpn Acad Ser B Phys Biol Sci. 2012;88:88–101.
- Chacko SM, Thambi PT, Kuttan R, Nishigaki I. Beneficial effects of green tea: a literature review. Chin Med. 2010;5:13.
- Ortiz-López L, Márquez-Valadez B, Gómez-Sánchez A, Silva-Lucero MDC, Torres-Pérez M, Téllez-Ballesteros RI, et al. Green tea compound epigallo-catechin-3-gallate (EGCG) increases neuronal survival in adult hippocampal neurogenesis in vivo and in vitro. Neuroscience. 2016;322:208–20.
- Pervin M, Unno K, Ohishi T, Tanabe H, Miyoshi N, Nakamura Y. Beneficial Effects of Green Tea Catechins on Neurodegenerative Diseases. Molecules [Internet]. 2018;23. Available from: http://dx.doi.org/10.3390/molecules23061297
- Babu PVA, Liu D. Green tea catechins and cardiovascular health: an update. Curr Med Chem. 2008;15:1840–50.
- Bhardwaj P, Khanna D. Green tea catechins: defensive role in cardiovascular disorders. Chin J Nat Med. 2013;11:345–53.
- Rains TM, Agarwal S, Maki KC. Antiobesity effects of green tea catechins: a mechanistic review. J Nutr Biochem. 2011;22:1–7.
- Hursel R, Westerterp-Plantenga MS. Catechin- and caffeine-rich teas for control of body weight in humans. Am J Clin Nutr. 2013;98:1682S – 1693S.
- Hursel R, Viechtbauer W, Westerterp-Plantenga MS. The effects of green tea on weight loss and weight maintenance: a meta-analysis. Int J Obes . 2009;33:956–61.
- Cooper R, Morré DJ, Morré DM. Medicinal benefits of green tea: part II. review of anticancer properties. J Altern Complement Med. 2005;11:639–52.
- Lambert JD. Does tea prevent cancer? Evidence from laboratory and human intervention studies. Am J Clin Nutr. 2013;98:1667S – 1675S.
- Park J-H, Bae J-H, Im S-S, Song D-K. Green tea and type 2 diabetes. Integr Med Res. 2014;3:4–10.
- Oliveira MR de, Nabavi SF, Daglia M, Rastrelli L, Nabavi SM. Epigallocatechin gallate and mitochondria-A story of life and death. Pharmacol Res. 2016;104:70–85.
- Schroeder EK, Kelsey NA, Doyle J, Breed E, Bouchard RJ, Loucks FA, et al. Green tea epigallocatechin 3-gallate accumulates in mitochondria and displays a selective antiapoptotic effect against inducers of mitochondrial oxidative stress in neurons. Antioxid Redox Signal. 2009;11:469–80.
- Most J, Timmers S, Warnke I, Jocken JW, van Boekschoten M, de Groot P, et al. Combined epigallocatechin-3-gallate and resveratrol supplementation for 12 wk increases mitochondrial capacity and fat oxidation, but not insulin sensitivity, in obese humans: a randomized controlled trial. Am J Clin Nutr. 2016;104:215–27.
- Jurgens TM, Whelan AM, Killian L, Doucette S, Kirk S, Foy E. Green tea for weight loss and weight maintenance in overweight or obese adults. Cochrane Database Syst Rev. 2012;12:CD008650.
- Baladia E, Basulto J, Manera M, Martínez R, Calbet D. [Effect of green tea or green tea extract consumption on body weight and body composition; systematic review and meta-analysis]. Nutr Hosp. 2014;29:479–90.
- Zhong X, Zhang T, Liu Y, Wei X, Zhang X, Qin Y, et al. Short-term weight-centric effects of tea or tea extract in patients with metabolic syndrome: a meta-analysis of randomized controlled trials. Nutr Diabetes. 2015;5:e160.
- Vázquez Cisneros LC, López-Uriarte P, López-Espinoza A, Navarro Meza M, Espinoza-Gallardo AC, Guzmán Aburto MB. Effects of green tea and its epigallocatechin (EGCG) content on body weight and fat mass in humans: a systematic review. Nutr Hosp. 2017;34:731–7.
- Hibi M, Takase H, Iwasaki M, Osaki N, Katsuragi Y. Efficacy of tea catechin-rich beverages to reduce abdominal adiposity and metabolic syndrome risks in obese and overweight subjects: a pooled analysis of 6 human trials. Nutr Res. 2018;55:1–10.
- Heber D. Pomegranate Ellagitannins. In: Benzie IFF, Wachtel-Galor S, editors. Herbal Medicine: Biomolecular and Clinical Aspects. Boca Raton (FL): CRC Press/Taylor & Francis; 2012.
- Ismail T, Calcabrini C, Diaz AR, Fimognari C, Turrini E, Catanzaro E, et al. Ellagitannins in Cancer Chemoprevention and Therapy. Toxins [Internet]. 2016;8. Available from: http://dx.doi.org/10.3390/toxins8050151
- Andreux PA, Blanco-Bose W, Ryu D, Burdet F, Ibberson M, Aebischer P, et al. The mitophagy activator urolithin A is safe and induces a molecular signature of improved mitochondrial and cellular health in humans. Nat Metab. 2019;1:595–603.
- Tan S, Yu CY, Sim ZW, Low ZS, Lee B, See F, et al. Pomegranate activates TFEB to promote autophagy-lysosomal fitness and mitophagy. Sci Rep. 2019;9:727.
- Ryu D, Mouchiroud L, Andreux PA, Katsyuba E, Moullan N, Nicolet-Dit-Félix AA, et al. Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nat Med. 2016;22:879–88.
- Torregrosa-García A, Ávila-Gandía V, Luque-Rubia AJ, Abellán-Ruiz MS, Querol-Calderón M, López-Román FJ. Pomegranate Extract Improves Maximal Performance of Trained Cyclists after an Exhausting Endurance Trial: A Randomised Controlled Trial. Nutrients [Internet]. 2019;11. Available from: http://dx.doi.org/10.3390/nu11040721
- Aviram M, Rosenblat M, Gaitini D, Nitecki S, Hoffman A, Dornfeld L, et al. Pomegranate juice consumption for 3 years by patients with carotid artery stenosis reduces common carotid intima-media thickness, blood pressure and LDL oxidation. Clin Nutr. 2004;23:423–33.
- Davidson MH, Maki KC, Dicklin MR, Feinstein SB, Witchger M, Bell M, et al. Effects of consumption of pomegranate juice on carotid intima-media thickness in men and women at moderate risk for coronary heart disease. Am J Cardiol. 2009;104:936–42.
- Basu A, Newman ED, Bryant AL, Lyons TJ, Betts NM. Pomegranate polyphenols lower lipid peroxidation in adults with type 2 diabetes but have no effects in healthy volunteers: a pilot study. J Nutr Metab. 2013;2013:708381.
- Li X-T, Chen R, Jin L-M, Chen H-Y. Regulation on energy metabolism and protection on mitochondria of Panax ginseng polysaccharide. Am J Chin Med. 2009;37:1139–52.
- Huang Y, Kwan KKL, Leung KW, Yao P, Wang H, Dong TT, et al. Ginseng extracts modulate mitochondrial bioenergetics of live cardiomyoblasts: a functional comparison of different extraction solvents. J Ginseng Res. 2019;43:517–26.
- Jin T-Y, Rong P-Q, Liang H-Y, Zhang P-P, Zheng G-Q, Lin Y. Clinical and Preclinical Systematic Review of Panax ginseng C. A. Mey and Its Compounds for Fatigue. Front Pharmacol. 2020;11:1031.
- Lee N, Lee S-H, Yoo H-R, Yoo HS. Anti-Fatigue Effects of Enzyme-Modified Ginseng Extract: A Randomized, Double-Blind, Placebo-Controlled Trial. J Altern Complement Med. 2016;22:859–64.
- Kim H-G, Cho J-H, Yoo S-R, Lee J-S, Han J-M, Lee N-H, et al. Antifatigue Effects of Panax ginseng C.A. Meyer: A Randomised, Double-Blind, Placebo-Controlled Trial. PLoS One [Internet]. Public Library of Science; 2013 [cited 2019 Nov 25];8. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3629193/
- Singh N, Bhalla M, de Jager P, Gilca M. An overview on ashwagandha: a Rasayana (rejuvenator) of Ayurveda. Afr J Tradit Complement Altern Med. 2011;8:208–13.
- Pratte MA, Nanavati KB, Young V, Morley CP. An alternative treatment for anxiety: a systematic review of human trial results reported for the Ayurvedic herb ashwagandha (Withania somnifera). J Altern Complement Med. 2014;20:901–8.
- Andrade C. Ashwagandha for anxiety disorders. World J. Biol. Psychiatry. 2009. p. 686–7.
- Zahiruddin S, Basist P, Parveen A, Parveen R, Khan W, Gaurav, et al. Ashwagandha in brain disorders: A review of recent developments. J Ethnopharmacol. 2020;257:112876.
- Lee D-H, Ahn J, Jang Y-J, Seo H-D, Ha T-Y, Kim MJ, et al. Withania somnifera Extract Enhances Energy Expenditure via Improving Mitochondrial Function in Adipose Tissue and Skeletal Muscle. Nutrients [Internet]. 2020;12. Available from: http://dx.doi.org/10.3390/nu12020431
- Lopresti AL, Smith SJ, Malvi H, Kodgule R. An investigation into the stress-relieving and pharmacological actions of an ashwagandha (Withania somnifera) extract: A randomized, double-blind, placebo-controlled study. Medicine . 2019;98:e17186.
- Salve J, Pate S, Debnath K, Langade D. Adaptogenic and Anxiolytic Effects of Ashwagandha Root Extract in Healthy Adults: A Double-blind, Randomized, Placebo-controlled Clinical Study. Cureus. 2019;11:e6466.
- Langade D, Kanchi S, Salve J, Debnath K, Ambegaokar D. Efficacy and Safety of Ashwagandha (Withania somnifera) Root Extract in Insomnia and Anxiety: A Double-blind, Randomized, Placebo-controlled Study. Cureus. 2019;11:e5797.
- Chandrasekhar K, Kapoor J, Anishetty S. A prospective, randomized double-blind, placebo-controlled study of safety and efficacy of a high-concentration full-spectrum extract of ashwagandha root in reducing stress and anxiety in adults. Indian J Psychol Med. 2012;34:255–62.
- Fuladi S, Emami SA, Mohammadpour AH, Karimani A, Manteghi AA, Sahebkar A. Assessment of Withania somnifera root extract efficacy in patients with generalized anxiety disorder: A randomized double-blind placebo-controlled trial. Curr Clin Pharmacol [Internet]. 2020; Available from: http://dx.doi.org/10.2174/1574884715666200413120413
- Shay KP, Moreau RF, Smith EJ, Smith AR, Hagen TM. Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential. Biochim Biophys Acta. 2009;1790:1149–60.
- Savitha S, Sivarajan K, Haripriya D, Kokilavani V, Panneerselvam C. Efficacy of levo carnitine and alpha lipoic acid in ameliorating the decline in mitochondrial enzymes during aging. Clin Nutr. 2005;24:794–800.
- Long J, Gao F, Tong L, Cotman CW, Ames BN, Liu J. Mitochondrial decay in the brains of old rats: ameliorating effect of alpha-lipoic acid and acetyl-L-carnitine. Neurochem Res. 2009;34:755–63.
- Liu J, Killilea DW, Ames BN. Age-associated mitochondrial oxidative decay: improvement of carnitine acetyltransferase substrate-binding affinity and activity in brain by feeding old rats acetyl-L- carnitine and/or R-alpha -lipoic acid. Proc Natl Acad Sci U S A. 2002;99:1876–81.
- Pershadsingh HA. Alpha-lipoic acid: physiologic mechanisms and indications for the treatment of metabolic syndrome. Expert Opin Investig Drugs. 2007;16:291–302.
- Chen W-L, Kang C-H, Wang S-G, Lee H-M. α-Lipoic acid regulates lipid metabolism through induction of sirtuin 1 (SIRT1) and activation of AMP-activated protein kinase. Diabetologia. 2012;55:1824–35.
- Carbonelli MG, Di Renzo L, Bigioni M, Di Daniele N, De Lorenzo A, Fusco MA. Alpha-lipoic acid supplementation: a tool for obesity therapy? Curr Pharm Des. 2010;16:840–6.
- Koh EH, Lee WJ, Lee SA, Kim EH, Cho EH, Jeong E, et al. Effects of alpha-lipoic Acid on body weight in obese subjects. Am J Med. 2011;124:85.e1–8.
- Li N, Yan W, Hu X, Huang Y, Wang F, Zhang W, et al. Effects of oral α-lipoic acid administration on body weight in overweight or obese subjects: a crossover randomized, double-blind, placebo-controlled trial. Clin Endocrinol . 2017;86:680–7.
- Breithaupt-Grögler K, Niebch G, Schneider E, Erb K, Hermann R, Blume HH, et al. Dose-proportionality of oral thioctic acid–coincidence of assessments via pooled plasma and individual data. Eur J Pharm Sci. 1999;8:57–65.
- Chowanadisai W, Bauerly KA, Tchaparian E, Wong A, Cortopassi GA, Rucker RB. Pyrroloquinoline quinone stimulates mitochondrial biogenesis through cAMP response element-binding protein phosphorylation and increased PGC-1alpha expression. J Biol Chem. 2010;285:142–52.
- Saihara K, Kamikubo R, Ikemoto K, Uchida K, Akagawa M. Pyrroloquinoline Quinone, a Redox-Active o-Quinone, Stimulates Mitochondrial Biogenesis by Activating the SIRT1/PGC-1α Signaling Pathway. Biochemistry. 2017;56:6615–25.
- Hwang P, Willoughby DS. Mechanisms Behind Pyrroloquinoline Quinone Supplementation on Skeletal Muscle Mitochondrial Biogenesis: Possible Synergistic Effects with Exercise. J Am Coll Nutr. 2018;37:738–48.
- Nakano M, Yamamoto T, Okamura H, Tsuda A, Kowatari Y. Effects of Oral Supplementation with Pyrroloquinoline Quinone on Stress, Fatigue, and Sleep. Functional Foods in Health and Disease. 2012;2:307–24.
- Harris CB, Chowanadisai W, Mishchuk DO, Satre MA, Slupsky CM, Rucker RB. Dietary pyrroloquinoline quinone (PQQ) alters indicators of inflammation and mitochondrial-related metabolism in human subjects. J Nutr Biochem. 2013;24:2076–84.
- Huxtable RJ. Physiological actions of taurine. Physiol Rev. 1992;72:101–63.
- Fakruddin M, Wei F-Y, Suzuki T, Asano K, Kaieda T, Omori A, et al. Defective Mitochondrial tRNA Taurine Modification Activates Global Proteostress and Leads to Mitochondrial Disease. Cell Rep. 2018;22:482–96.
- Hansen SH, Andersen ML, Cornett C, Gradinaru R, Grunnet N. A role for taurine in mitochondrial function. J Biomed Sci. 2010;17 Suppl 1:S23.
- Hansen SH, Andersen ML, Birkedal H, Cornett C, Wibrand F. The important role of taurine in oxidative metabolism. Adv Exp Med Biol. 2006;583:129–35.
- Jong CJ, Azuma J, Schaffer S. Mechanism underlying the antioxidant activity of taurine: prevention of mitochondrial oxidant production. Amino Acids. 2012;42:2223–32.
- Schaffer S, Kim HW. Effects and Mechanisms of Taurine as a Therapeutic Agent. Biomol Ther . 2018;26:225–41.
- Ripps H, Shen W. Review: taurine: a “very essential” amino acid. Mol Vis. 2012;18:2673–86.
- Maleki V, Mahdavi R, Hajizadeh-Sharafabad F, Alizadeh M. The effects of taurine supplementation on oxidative stress indices and inflammation biomarkers in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial. Diabetol Metab Syndr. 2020;12:9.
- Rosa FT, Freitas EC, Deminice R, Jordão AA, Marchini JS. Oxidative stress and inflammation in obesity after taurine supplementation: a double-blind, placebo-controlled study. Eur J Nutr. 2014;53:823–30.
- Xiao C, Giacca A, Lewis GF. Oral taurine but not N-acetylcysteine ameliorates NEFA-induced impairment in insulin sensitivity and beta cell function in obese and overweight, non-diabetic men. Diabetologia. 2008;51:139–46.
- Waldron M, Patterson SD, Tallent J, Jeffries O. The Effects of an Oral Taurine Dose and Supplementation Period on Endurance Exercise Performance in Humans: A Meta-Analysis. Sports Med. 2018;48:1247–53.
- Mahmoud AM, Hernández Bautista RJ, Sandhu MA, Hussein OE. Beneficial Effects of Citrus Flavonoids on Cardiovascular and Metabolic Health. Oxid Med Cell Longev. 2019;2019:5484138.
- Kumar A, Prakash A, Dogra S. Naringin alleviates cognitive impairment, mitochondrial dysfunction and oxidative stress induced by D-galactose in mice. Food Chem Toxicol. 2010;48:626–32.
- Alam MA, Subhan N, Rahman MM, Uddin SJ, Reza HM, Sarker SD. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv Nutr. 2014;5:404–17.
- Testai L, Piragine E, Piano I, Flori L, Da Pozzo E, Miragliotta V, et al. The Citrus Flavonoid Naringenin Protects the Myocardium from Ageing-Dependent Dysfunction: Potential Role of SIRT1. Oxid Med Cell Longev. 2020;2020:4650207.
- Kicinska A, Jarmuszkiewicz W. Flavonoids and Mitochondria: Activation of Cytoprotective Pathways? Molecules [Internet]. 2020;25. Available from: http://dx.doi.org/10.3390/molecules25133060
- Overdevest E, Wouters JA, Wolfs KHM, van Leeuwen JJM, Possemiers S. Citrus Flavonoid Supplementation Improves Exercise Performance in Trained Athletes. J Sports Sci Med. 2018;17:24–30.
- Martínez-Noguera FJ, Marín-Pagán C, Carlos-Vivas J, Rubio-Arias JA, Alcaraz PE. Acute Effects of Hesperidin in Oxidant/Antioxidant State Markers and Performance in Amateur Cyclists. Nutrients [Internet]. 2019;11. Available from: http://dx.doi.org/10.3390/nu11081898
- Calabrese EJ, Mattson MP, Calabrese V. Resveratrol commonly displays hormesis: occurrence and biomedical significance. Hum Exp Toxicol. 2010;29:980–1015.
- Mizuguchi Y, Hatakeyama H, Sueoka K, Tanaka M, Goto Y-I. Low dose resveratrol ameliorates mitochondrial respiratory dysfunction and enhances cellular reprogramming. Mitochondrion. 2017;34:43–8.
- Timmers S, Konings E, Bilet L, Houtkooper RH, van de Weijer T, Goossens GH, et al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 2011;14:612–22.
- Goh KP, Lee HY, Lau DP, Supaat W, Chan YH, Koh AFY. Effects of resveratrol in patients with type 2 diabetes mellitus on skeletal muscle SIRT1 expression and energy expenditure. Int J Sport Nutr Exerc Metab. 2014;24:2–13.
- Alway SE, McCrory JL, Kearcher K, Vickers A, Frear B, Gilleland DL, et al. Resveratrol Enhances Exercise-Induced Cellular and Functional Adaptations of Skeletal Muscle in Older Men and Women. J Gerontol A Biol Sci Med Sci. 2017;72:1595–606.
- Polley KR, Jenkins N, O’Connor P, McCully K. Influence of exercise training with resveratrol supplementation on skeletal muscle mitochondrial capacity. Appl Physiol Nutr Metab. 2016;41:26–32.
- de Oliveira MR, Nabavi SM, Braidy N, Setzer WN, Ahmed T, Nabavi SF. Quercetin and the mitochondria: A mechanistic view. Biotechnol Adv. 2016;34:532–49.
- Davis JM, Murphy EA, Carmichael MD, Davis B. Quercetin increases brain and muscle mitochondrial biogenesis and exercise tolerance. Am J Physiol Regul Integr Comp Physiol. 2009;296:R1071–7.
- Pelletier DM, Lacerte G, Goulet EDB. Effects of quercetin supplementation on endurance performance and maximal oxygen consumption: a meta-analysis. Int J Sport Nutr Exerc Metab. 2013;23:73–82.
- Kressler J, Millard-Stafford M, Warren GL. Quercetin and endurance exercise capacity: a systematic review and meta-analysis. Med Sci Sports Exerc. 2011;43:2396–404.
- Ou Q, Zheng Z, Zhao Y, Lin W. Impact of quercetin on systemic levels of inflammation: a meta-analysis of randomised controlled human trials. Int J Food Sci Nutr. 2020;71:152–63.
- Mohammadi-Sartang M, Mazloom Z, Sherafatmanesh S, Ghorbani M, Firoozi D. Effects of supplementation with quercetin on plasma C-reactive protein concentrations: a systematic review and meta-analysis of randomized controlled trials. Eur J Clin Nutr. 2017;71:1033–9.
- Tabrizi R, Tamtaji OR, Mirhosseini N, Lankarani KB, Akbari M, Heydari ST, et al. The effects of quercetin supplementation on lipid profiles and inflammatory markers among patients with metabolic syndrome and related disorders: A systematic review and meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr. 2020;60:1855–68.
- Guo W, Gong X, Li M. Quercetin Actions on Lipid Profiles in Overweight and Obese Individuals: A Systematic Review and Meta-Analysis. Curr Pharm Des. 2019;25:3087–95.
- Sahebkar A. Effects of quercetin supplementation on lipid profile: A systematic review and meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr. 2017;57:666–76.
- Tamtaji OR, Milajerdi A, Dadgostar E, Kolahdooz F, Chamani M, Amirani E, et al. The Effects of Quercetin Supplementation on Blood Pressures and Endothelial Function Among Patients with Metabolic Syndrome and Related Disorders: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Curr Pharm Des. 2019;25:1372–84.
- Serban M-C, Sahebkar A, Zanchetti A, Mikhailidis DP, Howard G, Antal D, et al. Effects of Quercetin on Blood Pressure: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J Am Heart Assoc [Internet]. 2016;5. Available from: http://dx.doi.org/10.1161/JAHA.115.002713
- Riva A, Ronchi M, Petrangolini G, Bosisio S, Allegrini P. Improved Oral Absorption of Quercetin from Quercetin Phytosome®, a New Delivery System Based on Food Grade Lecithin. Eur J Drug Metab Pharmacokinet. 2019;44:169–77.
- Pizzorno J. Glutathione! Integr Med . 2014;13:8–12.
- Atkuri KR, Mantovani JJ, Herzenberg LA, Herzenberg LA. N-Acetylcysteine–a safe antidote for cysteine/glutathione deficiency. Curr Opin Pharmacol. 2007;7:355–9.
- Pendyala L, Creaven PJ. Pharmacokinetic and pharmacodynamic studies of N-acetylcysteine, a potential chemopreventive agent during a phase I trial. Cancer Epidemiol Biomarkers Prev. 1995;4:245–51.
- Faghfouri AH, Zarezadeh M, Tavakoli-Rouzbehani OM, Radkhah N, Faghfuri E, Kord-Varkaneh H, et al. The effects of N-acetylcysteine on inflammatory and oxidative stress biomarkers: A systematic review and meta-analysis of controlled clinical trials. Eur J Pharmacol. 2020;884:173368.
- Polyak E, Ostrovsky J, Peng M, Dingley SD, Tsukikawa M, Kwon YJ, et al. N-acetylcysteine and vitamin E rescue animal longevity and cellular oxidative stress in pre-clinical models of mitochondrial complex I disease. Mol Genet Metab. 2018;123:449–62.
- Aparicio-Trejo OE, Reyes-Fermín LM, Briones-Herrera A, Tapia E, León-Contreras JC, Hernández-Pando R, et al. Protective effects of N-acetyl-cysteine in mitochondria bioenergetics, oxidative stress, dynamics and S-glutathionylation alterations in acute kidney damage induced by folic acid. Free Radic Biol Med. 2019;130:379–96.
- Sandhir R, Sood A, Mehrotra A, Kamboj SS. N-Acetylcysteine reverses mitochondrial dysfunctions and behavioral abnormalities in 3-nitropropionic acid-induced Huntington’s disease. Neurodegener Dis. 2012;9:145–57.
- Wright DJ, Renoir T, Smith ZM, Frazier AE, Francis PS, Thorburn DR, et al. N-Acetylcysteine improves mitochondrial function and ameliorates behavioral deficits in the R6/1 mouse model of Huntington’s disease. Transl Psychiatry. 2015;5:e492.




